The rays produced in a cathode tube are

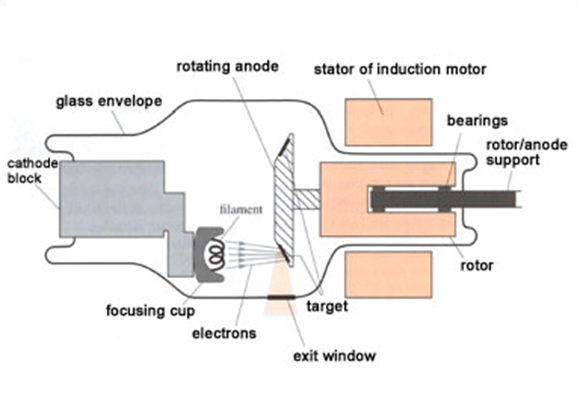
Of the X-rays and the greater is their penetrating power. The higher the voltage used, the shorter is the avelength The electrons are speeded up in the usual way, by applying a high p.d. The spiral is surrounded by a short metal cylinder which serves to focus the cathode rays on to the anticathode. In this tube the cathode takes the Gas-filled X-ray tubeįorm of a spiral of tungsten wire which, when heated by an electric current, gives out a stream of electrons by a process called thermionic emission (page 516). Modern tubes are based on a method devised by W. These rays are used in crime investigation.For reasons which cannot be discussed here the wavelength of the X-rays produced by the early gas tubes was difficult to control. In metal industry, these rays are used to detect cracks in structure and blow holes in metals.Ĥ. Mainly in hospital, to detect internal organism of infected part, X-rays are used.ģ. These rays are widely used in scientific researches and industries of medicine.Ģ. X-rays are very harmful to the human body if the body is exposed to X-rays for a long time.ġ. Even X-rays can penetrate through matter, which are opaque to the visible light.ħ. X-rays ionise the gas, when they pass through the gas.Ħ. When X-rays fall on zinc sulphide, they cause fluorescence.ĥ. As X-rays are not charged particles, so they are not deviated by an electric and magnetic field.Ĥ. X-rays travel with the velocity of light.ģ. X-rays travel always in straight line path.Ģ. When cathode rays hit this target T, then X-rays are produced.ġ. That target is made up of a heavy metal like molybdenum. The emitted cathode rays hit the target which is inclined at 45degrees to the path of cathode rays. E1 and E2 are the electromagnetic plates and their function is to control the acceleration of cathode rays emitted from cathode C. The cathode filament(C) which is connected to low tension voltage(L.T.). X-rays are mainly produced using coolidge tube. The range of wavelength of x-rays is 1Angstrom to 100Angstrom When highly energetic electrons are abruptly stopped by a metal target, then the kinetic energy of electrons lost manifests as electromagnetic radiation of low wavelength of order 1 Angstrom. When he was investigating about discharge of electricity through rarefied gases, he observed a fluorescence on a screen coated with Barium platino cyanide due to some invisible radiation called as X-rays. X-rays were discovered by German scientist Roentgen. Cathode rays have a mass of 1/1840 times the mass of Hydrogen atom. The velocity of cathode rays is 1/10 part of velocity of light.ĩ. Cathode rays are made up of fast moving electrons. When these rays strike a heavy solid metal, then X-rays are produced.Ĩ.

Cathode rayscan penetrate through thin foils, i.e., metal foils.ħ.

Therefore these rays are known as stream of electrons.Ħ. This shows that cathode rays carry negative charges. These rays deflects towards the positive electric field. This proves that the cathode rays consists of particles.ĥ.Cathode rays are also deflected by a electric field.
Cathode rays are deflected by a magnetic field. When these rays are focused on diamonds, they exhibit all colors depending on the quality of diamond.Ĥ. When cathode rays are focused on substances like Zinc Sulphide, a green visible light is observed. These rays have sufficient amount of kinetic energy such that A mica vane paddle wheel placed on the path of rays will be rotated.ģ. These rays leave perpendicular to the surface of the cathode.Ģ. Cathode rays always travel in straight line and they cast shadows. The pressure of enclosed gas in the tube can be varied by connecting it to vacuum pump.Ĭathode rays can be produced when the pressure inside the discharge tube is nearly 0.01mm of Hg.ġ. Another electrode connected to negative potential is called cathode. One electrode connected to positive potential is called anode. This tube has two electrodes at the ends. The pressure inside this tube can be varied for requirement conditions. Discharge tube This is tube made up of glass which is used to study the conduction of electricity through gases.
